Financial
Locations and Websites
The regulatory approval status and availability of Terumo Group products differs by country and region. You can receive product and service information from the Terumo Group company operating in the country or region where you will use the products or services.
North America
North America
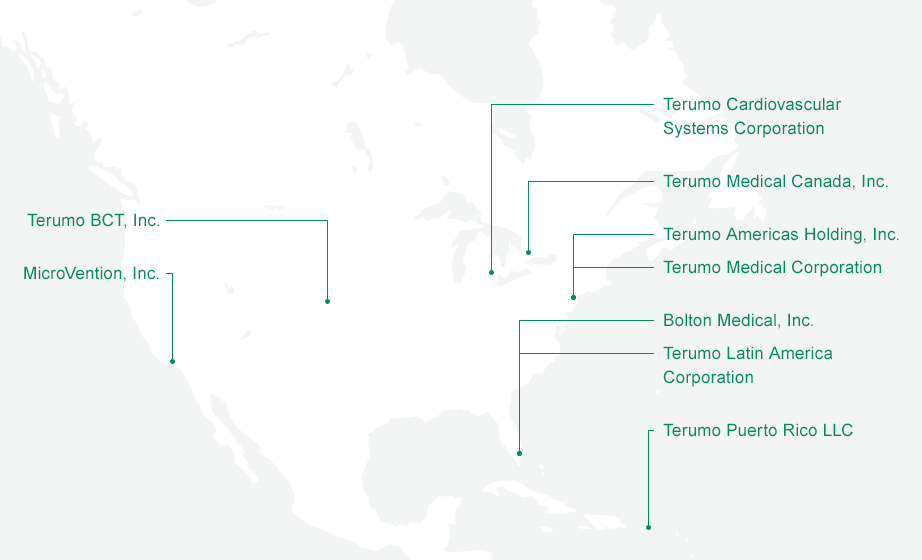
Terumo has been doing business in North America for nearly half a century. Terumo America Inc. (now Terumo Americas Holdings) was founded in 1971, and since then the Terumo business has grown much larger and more diverse. The Blood Management Company and the Neurovascular and Cardiovascular divisions of Terumo all now have their headquarters in North America, which is not only a center of Terumo research and development and production, but also the largest market that Terumo serves outside Japan, accounting for around 35% of total Terumo Group sales.
Headquartered on the East Coast of the United States, in New Jersey, Terumo Medical Corporation is the central location for four divisions, notably Terumo Health Outcomes (THO), Terumo Medical Products (TMP), the Pharmaceutical Solutions Division (PSD), and Terumo Interventional Systems (TIS). Located in Vaughan, Ontario Canada, Terumo Medical Canada Inc. serves the Canadian market. There are two manufacturing organizations within TMC. They are located in Elkton Maryland and Caguas Puerto Rico.
Terumo Cardiovascular Systems Corporation, headquartered in Michigan, handles the research, development, and production of Cardiovascular Business products.
Terumo Cardiovascular Systems Corporation, headquartered in Michigan, handles the research, development, and production of Cardiovascular Business products.
Headquartered on the West Coast in California, MicroVention Inc. develops, manufactures, and sells the neurovascular treatment devices of the Neurovascular Business. In recent years, North America has also become a Terumo center of device innovation, with the addition to the Terumo Group of Kalila Medical, a vascular closure device manufacturer; and of Bolton Medical , a Florida-based developer and manufacturer of stent grafts in the Vascular Graft business.
Terumo BCT and our Blood Management company are headquartered in the Western state of Colorado. The Blood Management business contributes to healthcare by providing products and services that sustain blood transfusions, blood and cell therapies, and related care in over 130 countries and regions globally.
South America
South America
EMEA
EMEA
Terumo Labeling Website
Click here for information on products manufactured by Terumo Corporation (legal manufacturer) in the EU and Australia, according to the respective regulations.
Greater China
Greater China
Asia and Pacific
Asia and Pacific
 Top
Top
Terumo (China) Holdings Co., Ltd.
Room 805, PICC Building 2
Jianguomenwai Street Chaoyang District Beijing, 100022 China
TEL
+86-10-6409-6685
FAX
+86-10-6409-6638
Terumo China (Hong Kong) Ltd.
5/F 80 Gloucester Road Wanchai Hong Kong
TEL
+852-2866-0811
FAX
+852-2529-0451
Terumo Medical (Shanghai) Co., Ltd.
Room1601-1602,T1,No.1388
Kaixuan Rd, Changning District
Shanghai 200051,China
TEL
+86-21-6237-1155
FAX
+86-21-6237-1150
Terumo Taiwan Medical Co., Ltd.
8th Floor, No. 4, Sec1, Zhongxiao W.Rd,
Zhongzheng Dist, Taipei City 100421, Taiwan
TEL
+886-2-2361-5123
FAX
+886-2-2361-5112
Terumo Medical Products (Hangzhou) Co., Ltd.
M4-9-5,
Economic & Technological Development Zone
Hangzhou 310018, China
TEL
+86-571-8731-8100
FAX
+86-571-8691-0293
Essen Technology (Beijing) Co., Ltd.
Building 1, NO.A12 Tianzhu West Road, Tianzhu Airport Industrial Zone A
Shunyi District, Beijing 101318, PR China
TEL
+86-10-6060-8836
FAX
+86-10-6060-8837
Wego Terumo (Weihai) Medical Products Co., Ltd.
3F, Gate 2, No.7 Weigao West Road
Torch Hi-Tech Science Park Weihai-City
Shandong Province 264210, China
TEL
+86-631-5716596
FAX
+86-631-5716578
