Financial
Locations and Websites
The regulatory approval status and availability of Terumo Group products differs by country and region. You can receive product and service information from the Terumo Group company operating in the country or region where you will use the products or services.
North America
North America
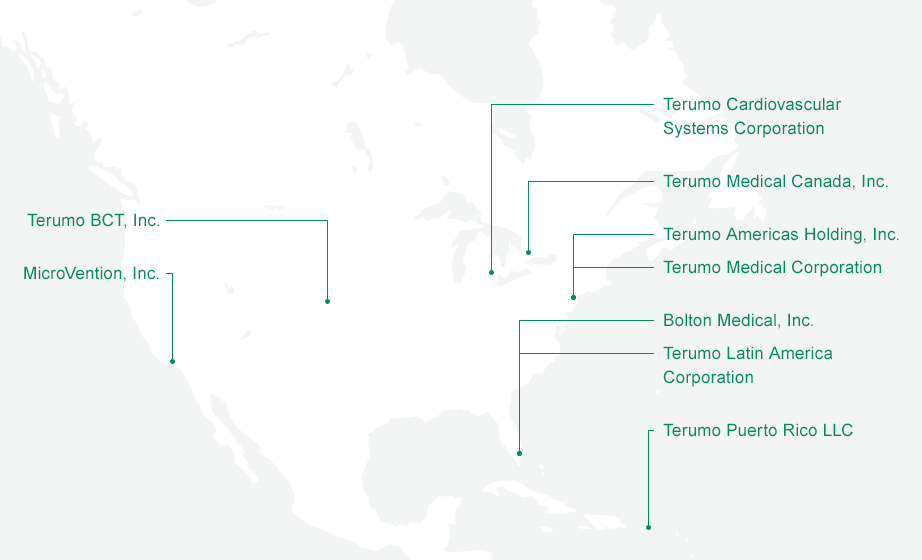
Terumo has been doing business in North America for nearly half a century. Terumo America Inc. (now Terumo Americas Holdings) was founded in 1971, and since then the Terumo business has grown much larger and more diverse. The Blood Management Company and the Neurovascular and Cardiovascular divisions of Terumo all now have their headquarters in North America, which is not only a center of Terumo research and development and production, but also the largest market that Terumo serves outside Japan, accounting for around 35% of total Terumo Group sales.
Headquartered on the East Coast of the United States, in New Jersey, Terumo Medical Corporation is the central location for four divisions, notably Terumo Health Outcomes (THO), Terumo Medical Products (TMP), the Pharmaceutical Solutions Division (PSD), and Terumo Interventional Systems (TIS). Located in Vaughan, Ontario Canada, Terumo Medical Canada Inc. serves the Canadian market. There are two manufacturing organizations within TMC. They are located in Elkton Maryland and Caguas Puerto Rico.
Terumo Cardiovascular Systems Corporation, headquartered in Michigan, handles the research, development, and production of Cardiovascular Business products.
Terumo Cardiovascular Systems Corporation, headquartered in Michigan, handles the research, development, and production of Cardiovascular Business products.
Headquartered on the West Coast in California, MicroVention Inc. develops, manufactures, and sells the neurovascular treatment devices of the Neurovascular Business. In recent years, North America has also become a Terumo center of device innovation, with the addition to the Terumo Group of Kalila Medical, a vascular closure device manufacturer; and of Bolton Medical , a Florida-based developer and manufacturer of stent grafts in the Vascular Graft business.
Terumo BCT and our Blood Management company are headquartered in the Western state of Colorado. The Blood Management business contributes to healthcare by providing products and services that sustain blood transfusions, blood and cell therapies, and related care in over 130 countries and regions globally.
South America
South America
EMEA
EMEA
Terumo Labeling Website
Click here for information on products manufactured by Terumo Corporation (legal manufacturer) in the EU and Australia, according to the respective regulations.
Greater China
Greater China
Asia and Pacific
Asia and Pacific
 Top
Top
Terumo Europe NV
Interleuvenlaan 40
3001 Leuven, Belgium
TEL
+32-16-38-12-11
FAX
+32-16-40-02-49
Terumo Italia S.r.l
Via Paolo di Dono 73 00142 Rome
Italy
TEL
+39-06-94-80-28-00
FAX
+39-06-60-51-32-33
Terumo Europe España SL
Calle Nanclares de Oca, 5, Parque Empresarial A2 Plaza,
Modulo 2d, 28806 Madrid, Spain
TEL
+34 902 10 12 98
FAX
+34 902 10 13 58
Terumo UK Ltd.
Building 2, Ground Floor Meadows Business Park Blackwater, Camberley,
Surrey, GU17 9AB, United Kingdom
TEL
+44-1276-480440
FAX
+44-1276-480465
Terumo Sweden AB
Sven Kallfelts Gata 18 SE-426 71 Vastra
Frolunda Sweden
TEL
+46-31-748-58-80
FAX
+46-31-748-58-90
Terumo France S.A.S.
Bâtiment Atria – 3ème étage, 1 avenue Edouard Belin,
92500 Rueil-Malmaison, France
Terumo (Deutschland) GmbH
Ludwig-Erhard-Strasse 6 65760
Eschborn Germany
TEL
+49-61-96-8023-0
FAX
+49-61-96-8023-200
Terumo Russia LLC
BC Northern Tower,
13th Floor 10 Testovskaya street
Moscow 123112, Russia
TEL
+7-495-988-4740
Terumo Middle East FZCO
Building 8WB – Office #317
Dubai Airport Free Zone (DAFZ)
P.O. Box 54614 Dubai
United Arab Emirates
TEL
+971-4-292-0200
FAX
+971-4-292-0222
Terumo South Africa Pty. Ltd.
Block 11 Pinewood Office Park, 33 Riley Road,
Woodmead, Gauteng, 2191, South Africa
TEL
+27 11 234 4515
Terumo Poland Sp.z o.o.
ul. 1 Sierpnia 6, Wisniowy Business Park,
02-134 Warsaw, Poland
TEL
+48 22 120 16 00
Terumo Regional Headquarter Company LLC
Samama Tower, Floor 5&6, King Fahad Road,
22222 Riyadh, Saudi Arabia
TEL
+27 11 234 4515
QUIREM Medical B.V.
Zutphenseweg 55, 3e verdieping,
7418AH Deventer, The Netherlands
TEL
+31 85 040 4170
Vascutek Ltd. (Terumo Aortic (UK))
Newmains Avenue Inchinnan Renfrewshire
PA4 9RR Scotland
TEL
+44-141-812 5555
FAX
+44-141-812 7170
Vascutek Deutschland GmbH
Luruper Chaussee 125
House 8 – Middle 22761 Hamburg Germany
TEL
+49-40-897133-0
FAX
+49-40-87408318
MicroVention Europe
30 bis rue du Vieil Abreuvoir 78100
Saint-Germain-en-Laye France
TEL
+33-1-39-21-5217
FAX
+33-1-39-21-1601
MicroVention Deutschland GmbH
Moskauer Str. 27
D-40227 Düsseldorf
Germany
TEL
+49-211-210-7980
FAX
+49-211-210-79829
MicroVention UK
Cobalt 13A, Cobalt Park
9 Silverfox Way
Newcastle upon Tyne, NE27 0QJ
United Kingdom
TEL
+44-191-258-6777
FAX
+44-191-258-5999
Terumo BCT Europe N.V.
Ikaroslaan 41 B- 1930 ZAVENTEM, Belgium
TEL
+32-2-715-0590
FAX
+32-2-715-0574
Terumo BCT, Ltd.
Old Belfast Road Millbrook Larne BT40
2SH United Kingdom
TEL
+44-1452-729292
FAX
+44-1452-729293
