Top
Top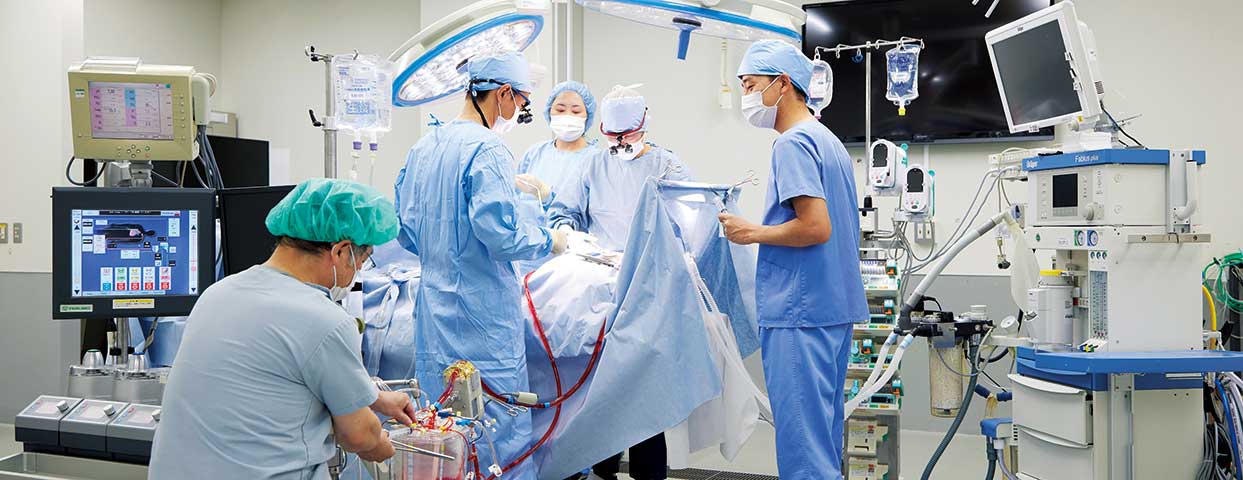
Terumo Cardiovascular
Terumo Cardiovascular develops and manufactures medical devices that function outside the body in place of the heart and lungs during cardiac surgery; oxygenators which perform blood oxygen exchange in place of the lungs; ECMO systems that support heart and lung function for patients in emergency care; and devices that help to support surgery without stopping the heart.
In collaboration with Terumo’s HeartSheet division, which has developed regenerative medicine products, the division is also contributing to better patient quality of life through innovative technologies for heart disease patients.
Terumo Cardiovascular
Note: The regulatory approval status and availability of these products differ by country or region.
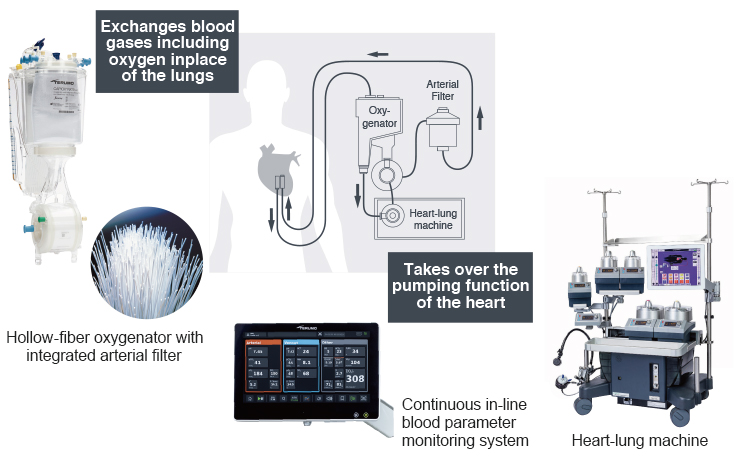
Cardiopulmonary Bypass System
Terumo offers devices that circulate blood in place of the heart and lungs during cardiovascular surgery in which the heart is stopped (“on pump” surgery).
During open-heart surgery, the heart-lung machine temporarily takes over the pumping function of the heart.
An oxygenator temporarily takes over the functions of the lungs, exchanging gases including oxygen and then returning oxygenated blood back to the patient via the cardiopulmonary bypass circuit.
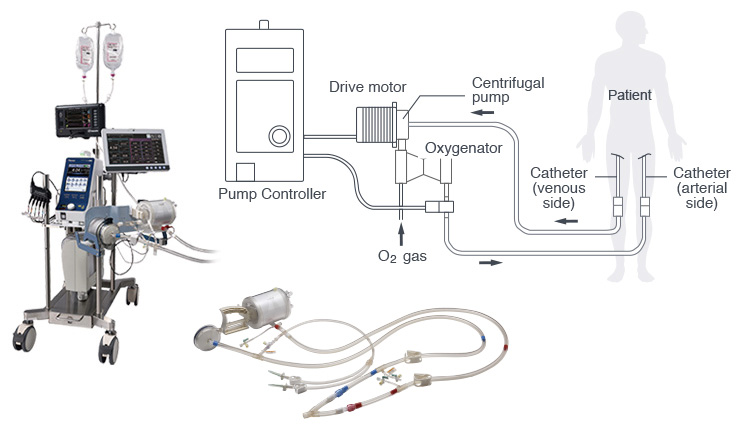
ECLS (extracorporeal life support system) /
ECMO (extracorporeal membrane oxygenation) system
Note: Commercially available only in Japan and some Asian countries.
This system performs the heart and lung functions for a patient with life-threatening respiratory or cardiac failure. This is done by inserting a catheter into a blood vessel of the thigh to bring blood outside the body, where it is cycled through a centrifugal pump and membrane oxygenator in a closed state.
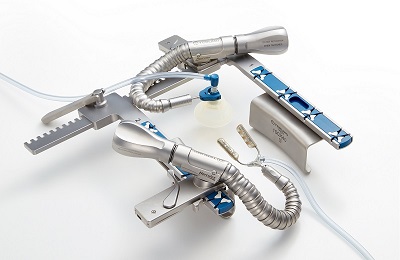
Off-Pump Coronary Artery Bypass Surgery Products
Beating Heart or Off-pump Coronary Artery Bypass Surgery avoids placing patients on-pump, and surgery is performed while the heart continues to beat. After chest incision, a stabilization system steadies only the portion of heart where the surgeon is working. This type of surgery is designed to avoid complications with on-pump procedures and benefits certain patient populations.
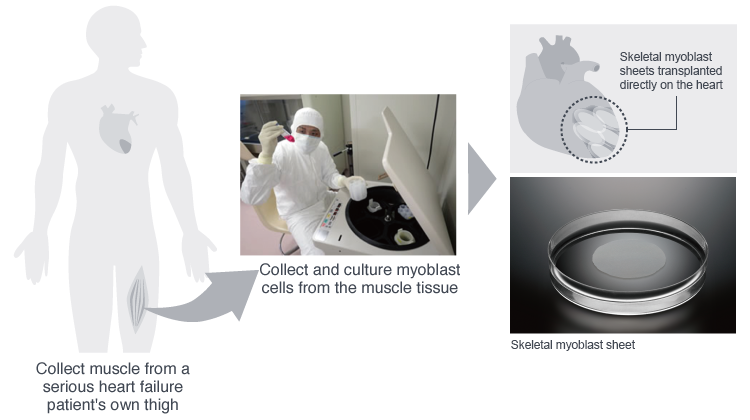
HeartSheet Division
Products for Regenerative Medicine
Individual skeletal myoblast sheets are made by taking skeletal myoblasts from the patient’s own thigh muscle, culturing them, and then forming them into sheets. These autologous skeletal myoblast sheets are transplanted directly onto the surface of the heart.